The Architecture of Electrodes: A Comprehensive Analysis of Electrochemical Components
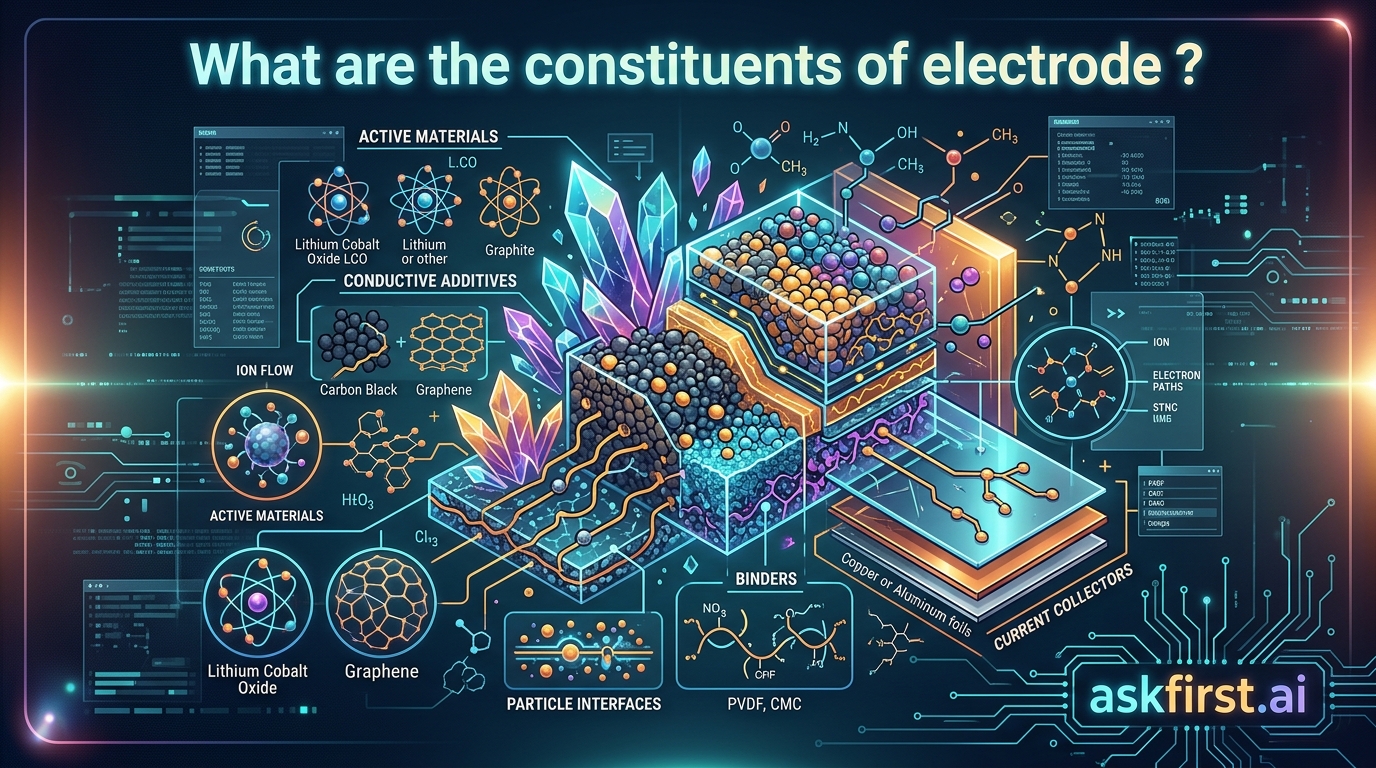
An electrode is the fundamental interface between an electronic circuit and an ionic medium, such as an electrolyte solution or a solid-state material. Whether utilized in a common household battery, a high-performance supercapacitor, or an industrial electrolysis cell, the performance of the entire electrochemical system is dictated by the precise composition and structural arrangement of the electrode. To understand the constituents of an electrode, one must examine the synergy between active materials, conductive additives, and polymeric binders, all of which are typically supported by a current collector.
1. The Active Material: The Engine of Electrochemical Potential
The active material is the primary component responsible for the storage or conversion of energy. In a lithium-ion battery, for example, the active material acts as a host for lithium ions. The chemistry of this material determines the theoretical capacity, voltage, and cycle life of the device.
- In Cathodes: Typically, metal oxides are used, such as Lithium Cobalt Oxide (LiCoO₂), Lithium Iron Phosphate (LiFePO₄), or Nickel Manganese Cobalt (NMC) oxides. These materials must facilitate reversible redox reactions.
- In Anodes: Graphite is the industry standard due to its layered structure that allows for the intercalation of lithium ions. However, emerging technologies utilize silicon-graphite composites or lithium titanate (LTO) to enhance energy density or power output.
- Expert Insight: As noted by Dr. M. Stanley Whittingham—a Nobel laureate and pioneer in battery chemistry—in his foundational research papers published in Science (1976), the intercalation mechanism is the bedrock of modern energy storage. The active material must possess high ionic conductivity and structural stability to withstand the mechanical stress of ion insertion and extraction.
2. Conductive Additives: Ensuring Electronic Percolation
While the active material is essential for energy storage, most active materials are inherently poor electronic conductors. To ensure that electrons can flow efficiently from the external circuit to the reaction sites, conductive additives are incorporated.
- Carbon Black and Super P: These are the most common additives, providing a high surface area and a robust network of conductive pathways.
- Carbon Nanotubes (CNTs) and Graphene: Modern high-performance cells utilize these nanostructures to create "long-range" conductive bridges, significantly reducing the internal resistance of the electrode.
- The Percolation Threshold: According to the principles of materials science outlined by W.D. Callister in Materials Science and Engineering: An Introduction, the conductive additive must be distributed in a specific ratio to reach a "percolation threshold," where the electrode becomes electronically conductive enough to sustain high current densities without significant heat loss (Joule heating).
3. Polymeric Binders: The Structural Adhesive
The binder acts as the "glue" that keeps the active material and conductive additives adhered to the current collector. Without a binder, the electrode would crumble due to the volume expansion and contraction that occurs during charge-discharge cycles.
- PVDF (Polyvinylidene Fluoride): This is the gold standard for many cathode systems, known for its excellent electrochemical stability and chemical resistance.
- CMC/SBR (Carboxymethyl Cellulose and Styrene-Butadiene Rubber): Often used in aqueous-processed anodes, these binders provide excellent flexibility and mechanical integrity, particularly in silicon-heavy electrodes that undergo significant swelling.
- Functionality: Beyond adhesion, the binder must be electrochemically inert within the operating voltage window of the battery to prevent parasitic side reactions that could degrade the electrolyte.
4. The Current Collector: The Electronic Backbone
The current collector serves as the mechanical support and the primary electronic highway. It is usually a thin metallic foil that connects the electrode material to the external terminals of the device.
- Aluminum Foil: Used almost exclusively for the cathode side because it forms a protective passivation layer at higher potentials, preventing corrosion.
- Copper Foil: Used for the anode side; it is chosen because it does not alloy with lithium at low potentials, maintaining structural stability.
- Design Considerations: The surface morphology of the foil is critical. As detailed in the Handbook of Battery Materials edited by Claus Daniel and Jürgen O. Besenhard, manufacturers often use "roughened" or "etched" foils to increase the surface contact area, thereby improving adhesion and reducing contact resistance between the slurry and the metal.
5. Electrolyte Interaction and Porosity
While not a solid "constituent" of the dry electrode, the pore structure created during the manufacturing process is a vital constituent of the functional electrode. During the manufacturing process, a slurry (a mixture of active material, binder, and additive) is coated onto the foil and dried. The remaining void space, or porosity, allows the liquid electrolyte to infiltrate the electrode. Without this porosity, ions would be unable to reach the reaction sites deep within the electrode, rendering the battery useless.
Conclusion
The electrode is not a simple block of material but a sophisticated composite system. It relies on the active material for storage, conductive additives for electron transport, binders for mechanical cohesion, and the current collector for external circuit integration. The optimization of these four constituents—often balanced at the nanoscale—is what enables the high-performance energy storage solutions that power modern electronics and electric vehicles. As research continues to evolve toward solid-state electrolytes and high-capacity anodes, the fundamental principles of these constituents remain the governing laws of electrochemical engineering.